- Endoch Medical is proudly 100% Canadian.
- info@endoch.com
- (905)-669-6999
Endoch Medical is proud to serve Canada as the only Canadian-owned and operating endoscopy OEM and independent repair facility. We proudly provide healthcare facilities across Canada with our very own line of Canadian endoscopes and GI supplies.





Ensure your endoscopic equipment performs reliably with quick turnaround times, detailed reports, and a warranty-backed repair service.
Endoch Medical is driven by a core commitment to patient safety through meticulous repairs and high-quality GI products and supplies. We understand the urgency within healthcare and minimize disruptions for our clients with efficient processes and rapid turnaround times.
Endoch Medical is proud to have served Canada as a 100% Canadian-owned and operating independent repair facility specializing in endoscopy for over 20 years, and we continue to expand our portfolio to better serve Canadian healthcare practitioners with a complete line of GI supplies and OEM capital equipment.

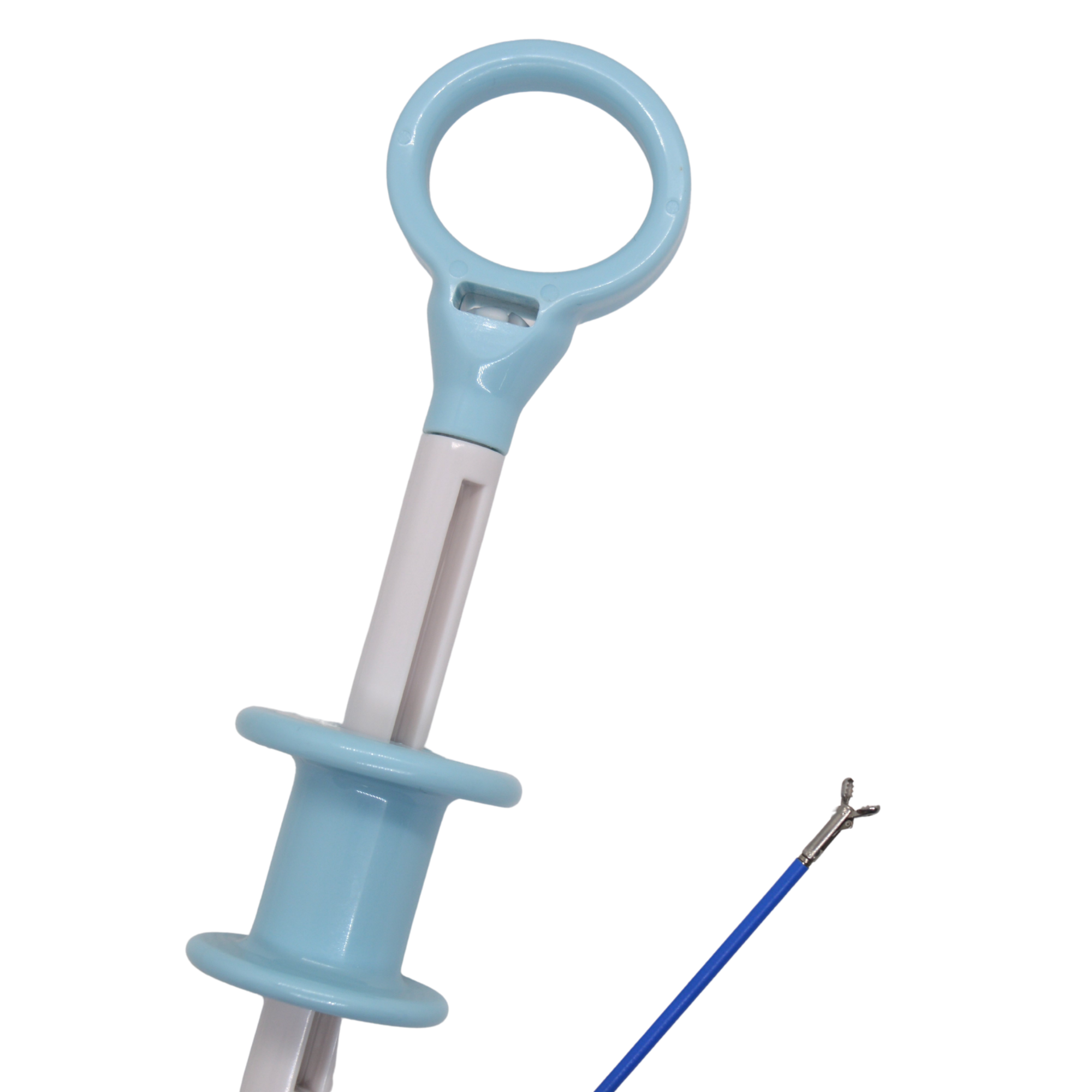

Trusted Quality & Efficiency: We prioritize patient safety with meticulous repairs and fast turnarounds, minimizing disruptions for your practice. we continue to expand our portfolio to better serve Canadian healthcare practitioners with a complete 100% Canadian line of GI supplies and OEM capital equipment.
Your Success is Ours: We’ve proven to our Canadian practitioners since 2006 that Canadian healthcare facilities benefit from alternative, competitive, Canadian service models right here at home. We build lasting partnerships through personalized service, clear communication, and competitive pricing.
Choosing Canadian: Endoch Medical is the only Canadian OEM Repair Facility operating within the GI healthcare space. We continuously improve our services and technologies to bridge OEM technical standards and discipline with the quality standards and accessibility of an independent service provider, all while maintaining certifications that well exceed other OEM and non-OEM providers in the Canadian healthcare space.
If you are interested in becoming a certified supplier for Endoch Medical, fill out a simple form today to get started.
Their recycling program has significantly reduced our waste and helped us stay compliant with regulations.

The field service team was prompt and professional. They fixed our ultrasound machine on-site, saving us a lot of hassle.

The endoscopy repair service was quick and efficient. Our equipment is working like new!

ISO 13485:2016 is the most internationally recognized quality management standard specific to medical devices. FDA regulations apply primarily to organizations that manufacture and distribute devices for American distribution.
100% Canadian OEM Repair Providers (like Endoch) don’t require this.
Automated Endoscope Reprocessor (AER) manufacturers commonly
state that they cannot guarantee devices serviced by other repair providers. These statements reflect weak liability boundaries, not automatic safety failures. AER manufacturers validate equipment under controlled conditions. They cannot validate every possible repair variation globally.
When a device is repaired anywhere, responsibility shifts from the OEM to the repair provider.
This does not invalidate reprocessing for the medical device in
question. All medical devices require confirmation that:
Healthcare facilities remain responsible for following the manufacturer’s IFU. Repair providers remain responsible for restoring the device to the condition that supports safe reprocessing. OEM manufacturers remains responsible for ensuring the device is manufactured to ensure safe reprocessing.
Safety is maintained when all parties uphold their responsibilities.
No. There are no changes to facility reprocessing workflows required when devices are properly restored to functional condition.
Facilities should continue to:
Endoch Medical supports this by ensuring the endoscope device
returned to you is suitable for those existing standard procedures.
Other OEMs do not sell repair parts to independent repair providers. For better or worse, this has resulted in Endoch Medical creating and using parts that now meet and sometimes exceed OEM minimum requirements and specifications.
It’s important to note that the decision to withhold repair parts is a commercial strategy, not a safety or legal requirement by other OEMs to do so. The critical factor is not branding – it is whether materials meet functional and safety requirements. Endoch Medical is MDSAP-certified. We perform our own stringent review of incoming parts, to confirm they match OEM quality, longevity, and functionality.
Market restriction limits competition, increases repair costs, and reduces service options for Canadian healthcare facilities overtime as alternative options begin to disappear from the market.
Endoch Medical is a 100% Canadian owned-and-operated MDSAP-certified endoscopy and GI supplies company. We meet all the Health Canada guidelines to manufacture, repair, and sell medical devices safely to Canadian healthcare facilities and practitioners.
MDSAP surpasses the minimum requirements of several international regulatory authorities, including Japan, Australia, Brazil, and even the US FDA, which means Endoch Medical exceeds the minimum expected requirements of an OEM manufacturer and an independent service provider on multiple international standards.
In short, you can trust us with your equipment.